Coligand modulated oxidative O-demethylation of a methyl ether appended tetradentate N-ligand in Co(ii) complexes†‡
Abstract
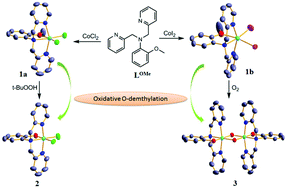
Two Co(II) complexes of the formula CoLOMeX2 (X = Cl− (1a); X = I− (1b)), where LOMe is 2-methoxy-N,N-bis(pyridin-2-ylmethyl) aniline, were synthesized and their structure, spectra and reactivity were studied. Upon oxidation of 1a and 1b, the ligand LOMe undergoes demethylation at the metal centre resulting in the formation of Co(III) complexes with modified phenoxide ligands. This is the very first example of oxidative O-demethylation reported at a Co(II) centre. The oxidative behaviour exhibits a striking dependence on the nature of coligands coordinated to the metal centre. The Co(II) complex 1a with stronger chloro coligands requires a strong oxidising agent like t-BuOOH for oxidative demethylation and the subsequent formation of a mononuclear Co(III) complex with a demethylated ligand, CoLO−Cl2 (2). On the other hand, complex 1b with weaker iodo coligands undergoes oxidation in the presence of the weak oxidant O2 to form a dihydroxo bridged binuclear Co(III) complex [Co2(LO−)2(OH)2]2+ (3) with modified phenoxide ligands. The oxidation of 1b to 3 is monitored and the intermediate Co(II) iodo aqua complex [CoLOMeI(H2O)]+ and Co(II) diaqua complex [CoLOMe(H2O)2]2+ are isolated and characterised.


 Please wait while we load your content...
Please wait while we load your content...