Coordination polymers of a bis-isophthalate bridging ligand with single molecule magnet behaviour of the CoII analogue†
Abstract
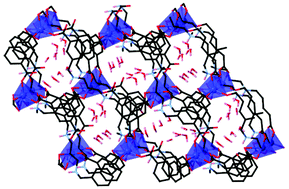
A linear diamine-bisisophthalate bridging linker N,N′-bis(1,3-dicarboxyphenyl-5-methylene)-1,3-dimethylpropanediamine, designed to incorporate amine/ammonium functionalities in the core of the ligand, has been isolated as the pentahydrate of its dihydrochloride salt (H6L)Cl2·5H2O. Using this compound, four new coordination polymers have been formed, namely poly-[M(H2L)]·4.5H2O (1M, where M = Co, Zn, Cd) and poly-[Cd(H2L)(OH2)]·DMF·7H2O (2). Compounds 1M are isostructural 2D coordination polymers that contain 1D channels occupied by water molecules. In the case of 1Co these form a well ordered hydrogen-bonding network as determined by single crystal X-ray studies. Compound 2, synthesised under similar conditions, is a 1D coordination polymer in which the metal is partially solvated. DC and AC magnetic studies of 1Co, which posseses a mononuclear cobalt(II) node, revealed single molecule magnet behaviour (SMM) with an effective barrier height Ueff of 37.7 K and τ0 = 1.02 × 10−9 s, among the highest reported for CoII coordination polymers.


 Please wait while we load your content...
Please wait while we load your content...