Oxidative C–N bond cleavage of (2-pyridylmethyl)amine-based tetradentate supporting ligands in ternary cobalt(ii)–carboxylate complexes†
Abstract
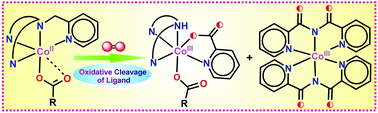
Three mononuclear cobalt(II)–carboxylate complexes, [(TPA)CoII(benzilate)]+ (1), [(TPA)CoII(benzoate)]+ (2) and [(iso-BPMEN)CoII(benzoate)]+ (3), of N4 ligands (TPA = tris(2-pyridylmethyl)amine and iso-BPMEN = N1,N1-dimethyl-N2,N2-bis((pyridin-2-yl)methyl)ethane-1,2-diamine) were isolated to investigate their reactivity toward dioxygen. Monodentate (η1) binding of the carboxylates to the metal centre favours the five-coordinate cobalt(II) complexes (1–3) for dioxygen activation. Complex 1 slowly reacts with dioxygen to enable the oxidative decarboxylation of the coordinated α-hydroxy acid (benzilate). Prolonged exposure of the reaction solution of 2 to dioxygen results in the formation of [(DPA)CoIII(picolinate)(benzoate)]+ (4) and [CoIII(BPCA)2]+ (5) (DPA = di(2-picolyl)amine and HBPCA = bis(2-pyridylcarbonyl)amide), whereas only [(DPEA)CoIII(picolinate)(benzoate)]+ (6) (DPEA = N1,N1-dimethyl-N2-(pyridine-2-ylmethyl)-ethane-1,2-diamine) is isolated from the final oxidised solution of 3. The modified ligand DPA (or DPEA) is formed via the oxidative C–N bond cleavage of the supporting ligands. Further oxidation of the –CH2– moiety to –C(![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O)– takes place in the transformation of DPA to HBPCA on the cobalt(II) centre. Labelling experiments with 18O2 confirm the incorporation of oxygen atoms from molecular oxygen into the oxidised products. Mixed labelling studies with 16O2 and H2O18 strongly support the involvement of water in the C–N bond cleavage pathway. A comparison of the dioxygen reactivity of the cobalt complexes (1–3) with those of several other five-coordinate mononuclear complexes [(TPA)CoII(X)]+/2+ (X = Cl, CH3CN, acetate, benzoylformate, salicylate and phenylpyruvate) establishes the role of the carboxylate co-ligands in the activation of dioxygen and subsequent oxidative cleavage of the supporting ligands by a metal–oxygen oxidant.
O)– takes place in the transformation of DPA to HBPCA on the cobalt(II) centre. Labelling experiments with 18O2 confirm the incorporation of oxygen atoms from molecular oxygen into the oxidised products. Mixed labelling studies with 16O2 and H2O18 strongly support the involvement of water in the C–N bond cleavage pathway. A comparison of the dioxygen reactivity of the cobalt complexes (1–3) with those of several other five-coordinate mononuclear complexes [(TPA)CoII(X)]+/2+ (X = Cl, CH3CN, acetate, benzoylformate, salicylate and phenylpyruvate) establishes the role of the carboxylate co-ligands in the activation of dioxygen and subsequent oxidative cleavage of the supporting ligands by a metal–oxygen oxidant.


 Please wait while we load your content...
Please wait while we load your content...