DFT study on the mechanism of bimetallic Pd–Zn-catalyzed cycloaddition of alkynyl aryl ethers with internal alkynes†
Abstract
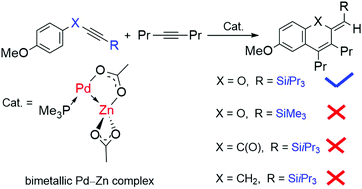
The reaction mechanism of bimetallic Pd–Zn-catalyzed cycloaddition of alkynyl aryl ethers with internal alkynes has been studied theoretically. Besides cycloaddition reaction, the dimerization of alkynyl aryl ethers was also considered. Both C6H5OC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3 and C6H5OC
CSiiPr3 and C6H5OC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiMe3 were chosen as the substrates. The reactions involve C–H activation of the substrate, acetic acid rotation, H transformation, MeC
CSiMe3 were chosen as the substrates. The reactions involve C–H activation of the substrate, acetic acid rotation, H transformation, MeC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CMe or substrate insertion into the Pd–phenyl bond and reductive elimination steps. It is found that cycloaddition is favored for C6H5OC
CMe or substrate insertion into the Pd–phenyl bond and reductive elimination steps. It is found that cycloaddition is favored for C6H5OC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3, while dimerization is preferred for C6H5OC
CSiiPr3, while dimerization is preferred for C6H5OC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiMe3, because the steric repulsion between two bulky SiiPr3 groups is relatively large and the steric repulsion between two small SiMe3 groups is relatively small. In addition, besides C6H5OC
CSiMe3, because the steric repulsion between two bulky SiiPr3 groups is relatively large and the steric repulsion between two small SiMe3 groups is relatively small. In addition, besides C6H5OC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3, four other substrates C6H5CH2C
CSiiPr3, four other substrates C6H5CH2C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3, C6H5C(O)C
CSiiPr3, C6H5C(O)C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3, C6H5SC
CSiiPr3, C6H5SC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3 and C6H5N(H)C
CSiiPr3 and C6H5N(H)C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3 have been calculated as the substrates for cycloaddition reaction with MeC
CSiiPr3 have been calculated as the substrates for cycloaddition reaction with MeC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CMe. Among the five substrates, C6H5OC
CMe. Among the five substrates, C6H5OC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3 has the lowest energy barrier (29.9 kcal mol−1), consistent with the experimental observation that C6H5OC
CSiiPr3 has the lowest energy barrier (29.9 kcal mol−1), consistent with the experimental observation that C6H5OC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CSiiPr3 is the appropriate substrate for successful cycloaddition.
CSiiPr3 is the appropriate substrate for successful cycloaddition.


 Please wait while we load your content...
Please wait while we load your content...