Coupled-substituted double-layer Aurivillius niobates: structures, magnetism and solar photocatalysis†
Abstract
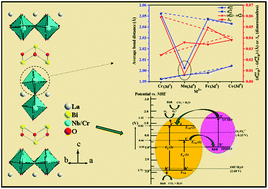
Development of layered perovskites for sunlight-driven catalysis has gained a lot of attention in contemporary inorganic materials research. While the compositional modifications of three-dimensional perovskites are ubiquitous, they are infrequent in the case of layered perovskites particularly with niobates when the perovskite layer thickness is low. We report here the solid state synthesis of a series of lead-free double-layer Aurivillius niobates, LaBi2Nb1.5M0.5O9 (M = Cr, Mn, Fe, Co), by adopting a heterovalent coupled substitution strategy with SrBi2Nb2O9. Rietveld structure refinements of the compounds using P-XRD data suggest the formation of 3dn transition metal incorporated double-layer Aurivillius niobates in the non-centrosymmetric A21am space group, isostructural with the parent. The compounds show optical absorption in the visible region with the absorption tail extending up to ∼650 nm and band gaps ranging from 2.25–2.94 eV. While the compounds show paramagnetic behaviour with no indication of magnetic phase transition or ordering in the temperature range 5–300 K, the Mn compound stabilizes with a low-spin (LS) configuration in contrast to all others (Cr, Fe and Co compounds), which adopt a high-spin (HS) configuration. The stabilization of the LS configuration (t42g) of the Mn compound occurs with eg → t2g electron redistribution due to the suppression of first-order Jahn–Teller (FOJT) distortion by the dominating second-order Jahn–Teller (SOJT) distortion of Nb5+ (4d0). The compounds exhibit photocatalytic rhodamine B degradation at pH 2 within 50–110 minutes under natural sunlight-irradiation and remain stable after five consecutive degradation cycles maintaining their activity largely intact. The heterovalent coupled substitution strategy adopted here will open up possibilities for transforming many other UV-active layered niobates into sunlight-active compounds without using toxic Pb or expensive Ag, while the paramagnetic nature of the compounds will be helpful in post-catalytic magnetic separation of the catalysts. It is believed that the electronic instability of the t32ge1g configuration of Mn due to competing FOJT and SOJT effects may have far-reaching consequences in modifying its magneto-structural and electron transport properties.


 Please wait while we load your content...
Please wait while we load your content...