Photoelectrochemical water-splitting over a surface modified p-type Cr2O3 photocathode†
Abstract
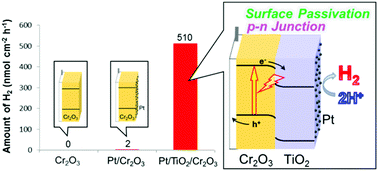
Cr2O3 is a p-type semiconductor with a negative conduction band minimum position suitable for photocathodic H2 generation. Therefore, Cr2O3 is a candidate photocathode material for photoelectrochemical (PEC) water-splitting. However, Cr2O3 has not yet been applied for the purpose of H2 generation because the efficiency and stability of the photocurrent generated by a Cr2O3 electrode are poor, due to high defect and vacancy concentrations. In the present work, the Cr2O3 surface was modified with n-type TiO2 after which Pt particles were deposited to catalyse H2 production. The TiO2 overlayer passivated the Cr2O3 surface states that otherwise cause deleterious interactions with the Pt particles. This layer also improved charge separation from the conduction band of Cr2O3 to the Pt co-catalyst, by forming a p–n junction. As a result of the TiO2 insertion, the cathodic photocurrent resulting from light absorption by Cr2O3 was enhanced and stabilized. This represents the first-ever use of Cr2O3 as a light-absorbing material in a multi-layered electrode to accomplish PEC water-splitting for H2 generation.
- This article is part of the themed collection: Metal Complexes and Inorganic Materials for Solar Fuel Production


 Please wait while we load your content...
Please wait while we load your content...