Methanol-assisted synthesis of Ni3+-doped ultrathin NiZn-LDH nanomeshes for boosted alkaline water splitting†
Abstract
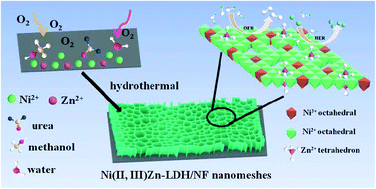
The construction of nanoporous structure combined with the optimization of electronic structure toward electrocatalysts could be a promising and effective approach to boosting their catalytic performance. Herein, we rationally synthesized a novel Ni3+-doped ultrathin NiZn layered double hydroxide nanomesh supported on nickel foam (Ni(II,III)Zn-LDH/NF-nm) by a facile one-step methanol-assisted hydrothermal method. Results show that methanol can not only trigger the generation of ultrathin nanomesh structure, but adjust portion of Ni2+ to Ni3+ and thus to result in the Ni3+-doped NiZn-LDH nanomesh material. The nanoporous feature endows Ni(II,III)Zn-LDH/NF-nm with abundant exposed catalytic active sites and fast mass transfer for alkaline water electrolysis. More importantly, the Ni3+ doping can facilitate the available formation of highly active NiOOH phase on the surface for the oxygen evolution reaction (OER), accompanied by increased oxygen vacancies that can greatly enhance the electronic conductivity, leading to the improved intrinsic activity and the accelerated electrocatalytic OER reaction kinetics. As expected, the as-prepared Ni(II,III)Zn-LDH/NF-nm has relatively low overpotentials of 320 and 370 mV to drive large current densities of 100 and 500 mA cm−2, respectively, and a small Tafel slope of 63.9 mV dec−1, extremely superior to RuO2/NF and NiZn-LDH/NF-ns counterpart. Meanwhile, the electrolyzer assembled for overall water splitting by Ni(II,III)Zn-LDH/NF-nm yields the outstanding catalytic activity and stability. This work highlights a feasible strategy to design and develop high-efficiency water splitting electrocatalysts via engineering on composition and nanostructure.


 Please wait while we load your content...
Please wait while we load your content...