Improved electrochemical performance of 2D accordion-like MnV2O6 nanosheets as anode materials for Li-ion batteries†
Abstract
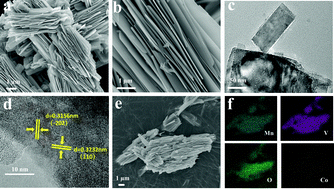
MnV2O6 is a promising anode material for lithium ion batteries with high theoretical specific capacity, abundant reserves and inexpensive constituent elements. However, in the process of lithization and de-lithization, the MnV2O6 anode material will form an amorphous phase, leading to collapse of its original layered structure; this greatly decreases its lithium storage capacity and specific capacity and affects its long-term cycle performance. In this study, 2D accordion-like MnV2O6 nanosheets with Co-doping are obtained via a hydrothermal route. The cobalt ions partially replace the positions of the manganese ions, and the emergence of Co3+ ions is inferred to induce the formation of a built-in electric field in the electrode to enhance the electrochemical behaviour of MnV2O6, presenting a high capacity of 1005.9 mA h g−1 after hundreds of cycles. The capacitive contribution to the total capacity is investigated to obtain insight into the kinetic analysis of its electrochemical behaviour. This study sheds light on an effective strategy to obtain excellent electrochemical behavior of MnV2O6-based materials and other transition metal oxides as electrodes for lithium storage.


 Please wait while we load your content...
Please wait while we load your content...