Altering the optoelectronic properties of boron difluoride formazanate dyes via conjugation with platinum(ii)-acetylides†
Abstract
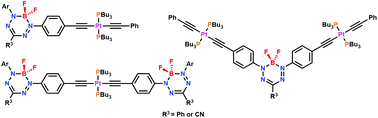
The combination of π-conjugated organic compounds and Pt(II)-acetylides is a powerful strategy for the production of functional optoelectronic materials. The presence of the heavy element, Pt, in these compounds enhances electronic delocalization generally resulting in low-energy absorption and emission maxima and often leads to intersystem crossing, resulting in phosphorescence. When boron complexes of N-donor ligands, such as boron dipyrromethenes (BODIPYs), are involved the molecular and polymeric materials produced have properties that are advantageous for their use as oxygen-sensors, in triplet–triplet annihilation, and as the functional components of photovoltaics. Based on these exciting results, we endeavored to thoroughly examine the effect of Pt(II)-acetylide conjugation on the properties of BF2 formazanate dyes, which offer improved redox properties and red-shifted absorption and emission bands compared to many structurally related BODIPYs. The results showed that phosphine-supported Pt(II)-acetylide incorporation enhanced electronic conjugation, rendering the electrochemical reduction of the BF2 formazanate dyes more difficult, while also red-shifting their absorption and emission maxima. Unlike similar BODIPYs, the presence of Pt(II) did not facilitate phosphorescence, but rather quenched fluorescence. This study provides significant insights into structure–property relationships and guiding principles for the design of BF2 formazanate dyes, a rapidly emerging family of readily accessible optoelectronic materials.
- This article is part of the themed collection: New Talent: Americas


 Please wait while we load your content...
Please wait while we load your content...