Tuning selectivity in nickel oxide-catalyzed oxidative dehydrogenation of ethane through control over non-stoichiometric oxygen density†
Abstract
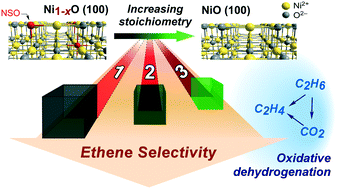
Despite bulk metal oxide non-stoichiometry often being recognized as a key determinant of catalytic performance, clarifying its catalytic function has remained elusive due in part to the highly complex nature of many catalyst surfaces. In this study, we demonstrate that the non-stoichiometric oxygen (NSO) density for thermally stable nickel oxide cubes can be manipulated without measurable changes in degree of crystallinity, particle size, and morphology. UV-vis spectroscopy, temperature programmed desorption, and H2-temperature programmed reduction analyses all evidence monotonic decreases in NSO density upon thermal treatment; crucially, these decreases persist upon exposure to ethane and oxygen under oxidative dehydrogenation of ethane (ODHE) reaction conditions. Such control over NSO density under reaction conditions is used to tune ODHE ethene selectivity; independent of reaction conditions, at isoconversion, ethene selectivity tracks qualitatively with NSO density. These trends in selectivity, however, do not extend to conditions resulting in higher fractional hydroxyl coverages, for example, in the presence of water co-feeds, where primary ODHE selectivities vary only at the highest treatment temperature used in our study. Overall, this study points to (i) the need for well-defined oxide materials that enable (ideally) the exclusive variation of a single physicochemical property, (ii) the importance of using multiple characterization techniques for quantifying various properties, and (iii) the sensitivity of selectivity trends to surface coverages of reaction intermediates prevalent during their measurement. For the purpose of elucidating structure–catalytic property relationships, the approach reported herein involving the use of well-defined thermally stable faceted oxide crystals has the potential to be broadly applicable within the field of bulk oxide catalysis.


 Please wait while we load your content...
Please wait while we load your content...