Adsorption driven formate reforming into hydride and tandem hydrogenation of nitrophenol to amine over PdOx catalysts†
Abstract
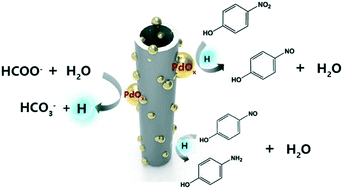
Due to their high toxicity and non-biodegradability, efficient reduction of nitroarenes to amines is of great practical importance, yet it still remains a significant challenge. Herein, we report PdO/PdO2 nanoparticles uniformly supported on titanate nanotubes (PdOx/TiNTs) for catalyzing the tandem dehydrogenation of sodium formate (SF) and hydrogenation of p-nitrophenol (PNP) to p-aminophenol (PAmP) under mild conditions. Notably, SF adsorption is mainly driven by the hydrogen bonding interactions between the H atom in SF and surface Pd sites, which factually makes the interface of PdOx/TiNT–SF an effective platform for C–H activation. Meanwhile, it is also found that the efficiency of the hydrogenation reaction depends on the reduction rate of the nitro group to nitroso group, and the O atoms adjacent to Pd are considered as the essential sites that facilitate this process. On the basis of the above two effects, the PdOx/TiNT catalyst shows unprecedented catalytic activity (turnover frequency, TOF, is 45.6 h−1) and good selectivity (∼100%) during PNP reduction at room temperature. This work deepens our understanding on tandem catalytic (de)hydrogenation systems, and will benefit the design of heterogeneous catalysts for the production of industrially important chemicals.


 Please wait while we load your content...
Please wait while we load your content...