Surface-state-induced upward band bending in P doped g-C3N4 for the formation of an isotype heterojunction between bulk g-C3N4 and P doped g-C3N4: photocatalytic hydrogen production†
Abstract
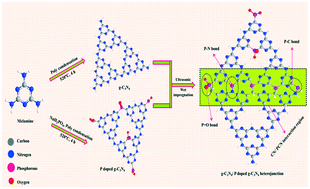
The staggered type heterojunction with g-C3N4 based nanomaterials has received much attention owing to its change in chemical potential between two semiconductors. As a result, the migration of charge carriers occurs via the interface electric field when the semiconductor band bends upward or downward. Herein, we made an isotype heterojunction between bulk g-C3N4 and bulk P–g-C3N4 by forming a surface P–C bond between them. The maximum hydrogen production of 1.6 mmol h−1 g−1 is obtained for the optimized heterojunction catalysts, which is 5.3 and 2.6 times higher than those of bulk g-C3N4 (0.30 mmol h−1 g−1) and bulk P–g-C3N4 (0.60 mmol h−1 g−1). Thus, the formation of surface P–C bonds hinders the charge carrier recombination on various parts of the heterojunction via enhancing the spatial charge separation and prolonged carrier lifetime. Besides, the altered electronic structure, interaction between g-C3N4 and P–g-C3N4, relocated work functions of P–g-C3N4 and distinct band edge positions were formed, which are responsible for the improved photocatalytic activity. Furthermore, the upward band bending of P–g-C3N4 and its higher delocalization ability led to higher conductivity and largely prevented electron–hole pair recombination.


 Please wait while we load your content...
Please wait while we load your content...