Oxygen vacancies enhance photocatalytic removal of NO over an N-doped TiO2 catalyst†
Abstract
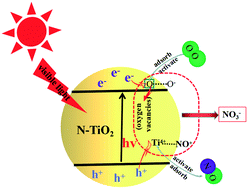
In this study, a non-metallic N-doped TiO2 photocatalyst (N–TiO2) was fabricated via facile co-deposition methods using titanium isopropoxide (Ti(OC3H7)4) and ammonia as precursors, and exhibited superior photocatalytic activity and selectivity to pure TiO2 on the degradation of NO under visible light irradiation at room temperature. The results of in situ diffuse reflectance infrared Fourier transform spectroscopy (in situ DRIFTS) suggested that NO was mainly adsorbed at Ti4+ sites, and then formed the NO+ active intermediates under visible light illumination. The co-adsorption of NO and molecular oxygen results indicated that the generation of the NO3− product over N–TiO2 was mainly attributed to the interaction between the NO species adsorbed on the catalyst surface and active oxygen (O−) rather than the surface hydroxyl. Moreover, the UV-vis diffuse reflectance spectra (DRS), photoluminescence (PL) and the photocurrent and electron paramagnetic resonance (EPR) results showed that TiO2 doped with N atoms not only enhanced the utilization efficiency of photo-generated charge carriers, but also promoted the formation of surface defects (e.g., oxygen vacancies) under visible light irradiation. Combined with X-ray photoelectron spectroscopy (XPS) and the O2 temperature-programmed desorption (O2-TPD) measurement analyses, it was found that oxygen vacancies not only facilitated the absorption and activation of molecular oxygen but also acted as a bridge between molecular oxygen and lattice oxygen in the catalyst (Ti4+ ↔ Ti3+). This resulted in an outstanding oxygen circulation between the Ti4+ and Ti3+ sites during the NO oxidation process under visible light irradiation. Thus, an enhanced NO removal efficiency and photocatalytic stability for NO oxidation occurred on N–TiO2.


 Please wait while we load your content...
Please wait while we load your content...