Particle size effect on the photocatalytic kinetics of barium titanate powders†
Abstract
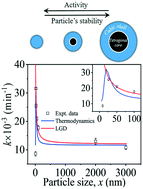
Particle size plays both a chemical and physical role in fields such as catalysis where a substantial surface-to-volume ratio is required in addition to photon utilization efficiency. Herein, we study the effect of particle size on the photocatalytic kinetics of barium titanate powders, and observe a sharp decrease of kinetics for nanoscale particles. We compare these experimental results to two models, namely the Landau–Ginzburg–Devonshire (LGD) and the thermodynamic model. By combining XRD and Raman spectroscopy to the macroscopic LGD model, we demonstrate that the smallest particle size to retain non-centrosymmetry and therefore a bulk photovoltaic contribution to the photocatalysis is in the range of 10 nm below which the surface-specific catalytic activity would be expected to decrease. The thermodynamic approach yields a maximum photoactivity for a particle size distribution between 15–25 nm, thus offering a framework to discriminate between both models.


 Please wait while we load your content...
Please wait while we load your content...