Untangling the active sites in the exposed crystal facet of zirconium oxide for selective hydrogenation of bioaldehydes†
Abstract
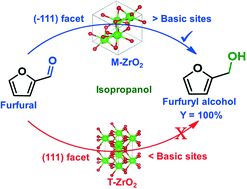
The present study reports the influence of the crystal phase, facets, and active sites of zirconium oxide (ZrO2) on the conversion of bio-aldehydes to their corresponding alcohols in isopropanol under mild reaction conditions. Various ZrO2-based catalysts, having different compositions of monoclinic and tetragonal crystal phases, are successfully prepared in the presence of a base via a solvothermal process. From the detailed characterization through XRD, TEM, CO2-TPD, XPS, AES, BET and poisoning studies, M-ZrO2-U-N, synthesized using zirconium oxynitrate and urea as a precursor and precipitant, respectively, in water, possesses a 100% monoclinic crystal phase with a maximum amount of exposed (−111) facets and surface oxygen concentration along with the highest number of basic sites. The catalytic study on the transformation of furfural (FFA) into furfuryl alcohol (FOH) reveals that M-ZrO2-U-N exhibits the best efficiency with a nearly quantitative yield of FOH. On the other hand, T-ZrO2-U-N, synthesized using zirconium oxynitrate and urea as a precursor and precipitant, respectively, in methanol, is found to have a 94.4% tetragonal phase and a 2.2-fold lower number of basic sites in comparison with M-ZrO2-U-N. The catalytic result with T-ZrO2-U-N displays the lowest activity in terms of the FOH yield (8.1%). According to the comparative and systematic catalytic studies with the various ZrO2 catalysts having different amounts of tetragonal and monoclinic phases, the ZrO2 catalyst having a more monoclinic phase with more exposed (−111) facets, basic sites, surface oxygen species and surface area is found to be crucial for the FFA conversion to FOH with high selectivity. M-ZrO2-U-N is found to be stable and recyclable and also shows excellent activity towards the transformation of other bio-aldehydes and ketones into their corresponding alcohols.


 Please wait while we load your content...
Please wait while we load your content...