Oxidative dehydrogenation of propane on silica-supported vanadyl sites promoted with sodium metavanadate†
Abstract
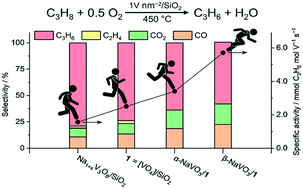
The promotion of silica-supported vanadyl species [VO4]/SiO2 (1) by α-NaVO3 or β-NaVO3 enhances the specific rate of the propene formation in oxidative dehydrogenation of propane (ODP) by, respectively, 30 and 125% at 450 °C and ca. 1 V nm−2 nominal coverage. The increased rate of propene formation is offset only moderately by a decreased selectivity to propene, which declines by 10 and 15% relative to 1 (74%) in α-NaVO3/1 and β-NaVO3/1, at 5.8 and 8.2% propane conversion. The structural characterization of the promoted catalysts by Raman mapping, X-ray absorption near edge structure (XANES), transmission electron microscopy (TEM) and solid-state nuclear magnetic resonance (51V and 23Na MAS NMR) allowed for associating the higher specific activity of β-NaVO3/1 with a higher dispersion of vanadium sites on the silica support, while the agglomeration of these sites with the concomitant formation of a poorly dispersed Na1+xV3O8 phase is related to a decreased catalytic activity. Surprisingly, solid-state 51V NMR and Raman spectroscopies reveal that the α-NaVO3/1 and β-NaVO3/1 catalysts contain the metastable β-NaVO3 phase, explained by a more favorable interaction of Na1+xV3O8/SiO2, formed after calcination in both materials, with β-NaVO3 than with α-NaVO3.


 Please wait while we load your content...
Please wait while we load your content...