Bimetallic Fe–Cu/beta zeolite catalysts for direct hydroxylation of benzene to phenol: effect of the sequence of ion exchange for Fe and Cu cations†
Abstract
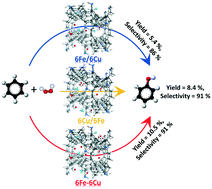
Recently, bimetallic cation-exchanged zeolite catalysts have received much attention. In this study, we focused on the bimetallic Fe and Cu beta zeolite, and investigated the effect of the ion-exchange sequence on the physicochemical and catalytic properties upon introducing Fe and Cu cations into beta zeolites. The bimetallic catalysts were prepared by the ion-exchange method using NH4+-type beta in three sequences, namely, 1.) Fe followed by Cu, 2.) Cu followed by Fe, and 3.) Fe and Cu at the same time. The bimetallic Fe–Cu/beta zeolites prepared by different sequences showed different physicochemical properties and catalytic properties in the hydroxylation of benzene with H2O2. To clarify the reasons for this, the content and states of the metallic species were systematically characterized by ICP-AES, in situ NO adsorption FTIR spectroscopy, XPS and H2-TPR technologies. It was indicated that the ion-exchange sequence influenced the content and the states of the Fe and Cu species, and that the metallic state had a greater influence on the catalytic properties than the content.


 Please wait while we load your content...
Please wait while we load your content...