Removal of toluene as a biomass tar surrogate by combining catalysis with nonthermal plasma: understanding the processing stability of plasma catalysis
Abstract
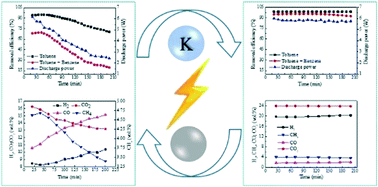
In this study, toluene (tar surrogate) removal under a simulated gasification gas (SGG) atmosphere was conducted by combining DBD plasma with a nickel catalyst. The processing stability of plasma catalysis was investigated in terms of toluene removal and SGG reactions, and some attempts at stability improvement including the addition of H2 or H2O and K-modification of the catalyst were conducted. The results indicate that plasma exerts great effects on the carbon deposition rate on the plasma-covered catalyst surface to affect the processing stability, and the effect, increasing or to some extent decreasing the carbon deposition rate, depends on the operating conditions and catalysts. In a Ni/γ-Al2O3 reactor with a SGG atmosphere and H2 or H2O addition, the higher carbon deposition rate induced by plasma leads to poor stability, while under the high H2O addition amount condition, the activation effect of plasma combined with the H2O/CO2 adsorption enhancement of K-modified Ni/γ-Al2O3 prevented the higher carbon deposition rate in the plasma-covered area and hence successfully improved the processing stability.


 Please wait while we load your content...
Please wait while we load your content...