Selective hydrogen production from formic acid decomposition over Mo carbides supported on carbon materials†
Abstract
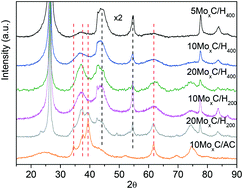
The decomposition of formic acid to obtain hydrogen has been studied using molybdenum carbides supported on activated carbon and two high surface area graphites, H200 (200 m2 g−1) and H400 (400 m2 g−1). Particular attention is paid to the effect of Mo loading. The catalysts were prepared in situ using a mixture of CH4 and H2 at a temperature of up to 700 °C. Under these conditions, carburization was mostly complete. We observed that the support influenced the MoxC phase obtained so that it seems that the ratio of defective carbon influences the phase. However, for these materials the C/Mo ratio did not influence the obtained crystal phase. Characterization by XRD showed that while the β-Mo2C phase was obtained over activated carbon and over H200, in contrast, MoOxCy was obtained over H400. These catalysts reached 100% conversion on formic acid decomposition at temperatures in the range of 190–250 °C and were also highly selective under these mild conditions, with values for CO2 selectivity in the range of 85.0–96.5%. The best results were achieved over a 10 wt% Mo loading on activated carbon that reached 96.5% selectivity to H2. Also, changes in the molybdenum phases were observed on the spent catalyst. Some redox transformations during reaction were responsible for the transformation of β-Mo2C into oxycarbide MoOxCy. In summary, the results of the catalytic performance indicated that the β-Mo2C phase was more active, selective and stable than MoOxCy under the studied conditions.


 Please wait while we load your content...
Please wait while we load your content...