In situ modulation of silica-supported MoO2/Mo2C heterojunction for enhanced hydrogen evolution reaction†
Abstract
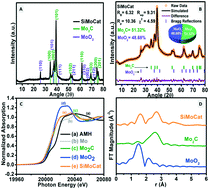
Hydrogen being a promising source of clean energy, the production of hydrogen using electrocatalysis and the development of carbon-neutral energy conversion technologies are crucial. Lately, electrocatalysts with multiple solid–solid heteroatom interfaces have been gaining importance due to their improved HER activity. Herein, a mesoporous composite of molybdenum carbide and molybdenum oxide on a silica platform (SiMoCat) was developed, which exhibited an overpotential of 71 mV to drive a current density of 10 mA cm−2, very close to the commercially available Pt/C (η10 = 60 mV), and a Tafel slope value of 35 mV dec−1 with an onset potential of 31 mV. Even after 3500 catalytic cycles, SiMoCat was found to be quite stable and did not show any significant change in the catalytic activity. SiMoCat was obtained by judicious variation in the catalyst precursor and the reaction conditions, resulting in a composition containing 51.32% Mo2C and 48.68% MoO2. Theoretical studies revealed that the integration of Mo2C and MoO2 heterostructure with SiO2 substrate enhances the surface charge distribution of the SiMoCat interface and assists the formation of an energetically active catalytic region. The values of ΔGH* on the C, Mo (∼1.9 eV, ⋍2.44 eV) and O, Mo (⋍0.54 eV, ⋍2.29 eV) sites of Mo2C/MoO2 of SiMoCat are optimized as the effect percolates through the nanolayers and hence the H* adsorption kinetics of the interface are better than those of the individual systems.


 Please wait while we load your content...
Please wait while we load your content...