Unraveling the hydrolysis of β-1,4-glycosidic bonds in cello-oligosaccharides over carbon catalysts†
Abstract
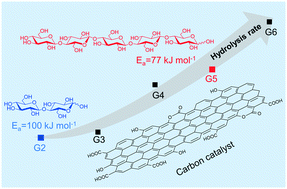
Carbon catalysts having weakly acidic groups are uniquely active for hydrolysis of cellulose to produce cello-oligosaccharides and glucose. Although adsorption of cellulose molecules on carbon is considered as the cause for this behavior, the effect of adsorption on the reaction is not well understood. In order to understand the underlying mechanism, we investigated the hydrolysis of cello-oligosaccharides with varying chain lengths over different catalysts. Carbon catalysts favored hydrolysis of larger oligosaccharides with an 11-fold increase in the reaction rate constant from cellobiose to cellohexaose. The activation energy required to cleave the glycosidic bonds was reduced concurrently with the increase in molecule size. Based on these data, in conjugation with the stronger affinity of adsorption for larger oligosaccharides, we propose that axial adsorption within the micropores of carbon causes conformational change in the structure of cello-oligosaccharide molecules, resulting in reduction of activation energy required to cleave the β-1,4-glycosidic bonds. Consequently, this translates to the higher rate of reaction for larger cello-oligosaccharides and explains the high reactivity of carbon catalysts towards cellulose hydrolysis.


 Please wait while we load your content...
Please wait while we load your content...