Effect of Sn-self diffusion via H2 treatment on low temperature activation of hematite photoanodes†
Abstract
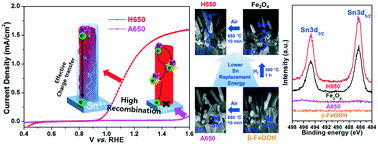
The objective of this study was to report the preparation of a highly efficient hematite nanorod photoanode by H2 treatment of the β-FeOOH nanorod with subsequent quenching in air at lower temperature activation for photoelectrochemical (PEC) water oxidation. The hematite nanorod photoanode (H650) prepared by thermally treating the β-FeOOH nanorod at 360 °C for 1 h under H2 flow and quenching at 650 °C for 10 min in air exhibited a remarkable photocurrent of 1.17 mA cm−2 at 1.23 V vs. RHE, which was 20 times higher than that of hematite quenched at the same temperature without H2 treatment. Such enhanced PEC performance was attributed to high Sn4+ diffusion from the fluorine doped SnO2 (FTO) substrate via H2 treatment and Sn4+ doping by subsequent lower temperature quenching. Our density functional theory (DFT) calculation supports our experimental results and proposed mechanism in that the Sn replacement reaction of magnetite (Fe3O4, H2-reduced β-FeOOH) occurs at a lower temperature and requires a smaller energy than that of akaganeite (β-FeOOH).


 Please wait while we load your content...
Please wait while we load your content...