Synergistic effect between gold nanoparticles and Fe-doped γ-MnO2 toward enhanced aerobic selective oxidation of ethanol†
Abstract
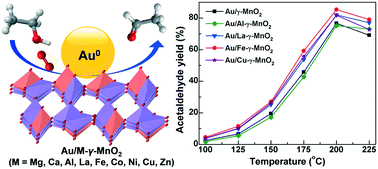
Developing synergistic catalysts between gold nanoparticles (AuNPs) and supports is crucial to enhance the catalytic efficiency of supported AuNP catalysts for the aerobic selective oxidation of ethanol to acetaldehyde (AC). In this study, for the first time, AuNPs supported on metal-doped γ-MnO2 (Au/M-γ-MnO2, M = Mg, Ca, Al, La, Fe, Co, Ni, Cu, Zn) catalysts with high gold dispersion were prepared by the colloidal deposition method and used in gas-phase ethanol oxidation to achieve up to 85% AC yield at 200 °C. It was found that the Au/M-γ-MnO2 catalysts show obviously higher reducibility and activity than the M-γ-MnO2 supports, and catalyst prereduction can further improve the catalytic activity. The strongest gold–support synergy was observed in the optimal Au/Fe-γ-MnO2 catalyst with a moderate Fe-doping amount, which can achieve the highest space–time-yield (∼3.2 g gcat−1 h−1) and outperform the previously reported supported AuNP catalysts at 200 °C. Kinetic isotope effect studies clearly suggest that the α-C–H cleavage is more rate-limiting than the O–H cleavage in ethanol oxidation and the surface adsorbed oxygen species on MnO2 is involved in the initial ethanol activation. By comparing the XPS spectra of the fresh and spent Au/Fe-γ-MnO2 catalysts, a novel Au–Mn–Fe synergy was proposed to account for its superior catalytic activity. The Mn2+ and Fe2+ defect-enriched catalyst interface is thought to facilitate the activation of O2 and ethanol at lower temperature.


 Please wait while we load your content...
Please wait while we load your content...