Highly efficient alloyed NiCu/Nb2O5 catalyst for the hydrodeoxygenation of biofuel precursors into liquid alkanes†
Abstract
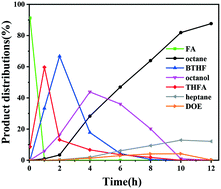
Hydrodeoxygenation (HDO) is a crucial process for the synthesis of biofuels from renewable biomass. Here, several bimetallic Ni–M/Nb2O5 catalysts (M = Fe, Co, Cu) were synthesized and evaluated in the HDO of biofuel precursors (aldol adduct of furfural with acetone) to liquid alkane and it was found that Ni–Cu/Nb2O5 has the best performance (86.5% yield of octane and 5.1% yield of heptane). Various characterization techniques show that Ni–Cu alloy is formed over the Ni–Cu/Nb2O5 catalyst, which may have the main active metal sites. Moreover, the Ni–Cu alloy has more sites for the adsorption–activation of H2, leading to the high activity. During the HDO process, the ring-opening of the intermediate butyl-tetrahydrofuran (BTHF) and the conversion of octanol are two rate-determining steps. The kinetic study confirms that the ring-opening of BTHF over Ni–Cu/Nb2O5 is more favorable than that over Ni/Nb2O5. More importantly, the Ni–Cu alloy significantly restrained the undesirable decarbonylation due to the weak adsorption of carbonyl, which was key for the efficient production of Cn alkanes. This work provides an in-depth understanding of the role of Ni–Cu alloys and new insights into the design of non-noble metal catalysts for the HDO of biofuel precursors.


 Please wait while we load your content...
Please wait while we load your content...