Facet-dependent activity of hematite nanocrystals toward the oxygen evolution reaction†
Abstract
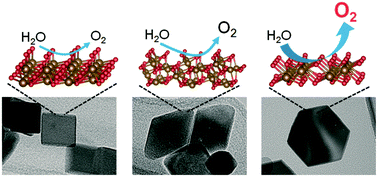
The electrocatalytic activity of hematite with different exposed facets toward the oxygen evolution reaction (OER) was investigated. Hematite nanoparticles with pseudo cubic, hexagonal bipyramid and plate morphologies were prepared, which predominantly have exposed (012), (113) and (001) facets, respectively. The OER activity of hematite measured under alkaline conditions was significantly dependent on the surface orientation, and the onset potential of the (001) facet was more than 130 mV more negative than those of the (012) and (113) facets. On the basis of the results of spectroelectrochemical measurements and theoretical calculations, this higher activity of the (001) facet was attributed to the lower energy required for the formation of oxo intermediate species. When pH conditions changed from alkaline to neutral, the facet dependence was less pronounced and the OER initiated from almost the same potential on all of the three facets. These findings indicate that the factor determining the OER activity of hematite differs depending on pH, and the surface structure strongly affects the OER activity under conditions where the concerted transfer of protons and electrons proceeds.


 Please wait while we load your content...
Please wait while we load your content...