0D/2D CeO2 quantum dot/NiO nanoplate supported an ultralow-content Pt catalyst for the efficient oxidation of formaldehyde at room temperature
Abstract
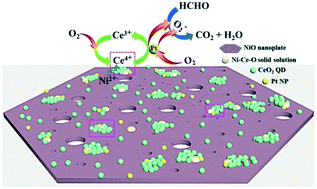
The development of cost-effective and high-powered catalysts to completely decompose formaldehyde (HCHO) into CO2 and water at room temperature is an important challenge. In this study, we employed a hydrothermal method, followed by an impregnation-chemical reduction, to successfully obtain the hybrid architecture of a 0D/2D CeO2 quantum dot (QD)/porous NiO hexagonal nanoplate supported ultralow-content (0.08 wt%) Pt. 0D/2D CeO2/NiO composite supported Pt (Pt/NiCe4) with Ce/Ni molar ratio of 1 : 6 showed an HCHO removal efficiency of 87% in an hour, which was significantly higher than that of Pt/CeO2 (56%) and Pt/NiO (54%). The improved catalytic activity of Pt/NiCe4 is due to the unique morphology of CeO2 QDs deposited over the NiO nanoplates, leading to more exposed active sites and close contact of the components on a large scale, and a large amount of the active surface oxygen species stemmed from the insertion of Ni into the CeO2 lattice and the crucial role of Pt in the activation of O2 from air. Moreover, the possible reaction mechanism for HCHO oxidation over Pt/NiCe4 was proposed based on the results of the in situ diffused reflectance infrared Fourier transform spectroscopic studies and the performance test. This study may provide a new enlightenment for constructing versatile catalysts for application in indoor air purification.


 Please wait while we load your content...
Please wait while we load your content...