Redox-switchable ring-opening polymerization by tridentate ONN-type titanium and zirconium catalysts†
Abstract
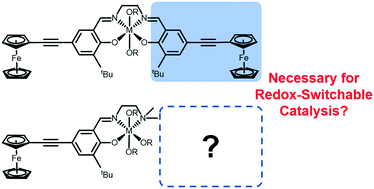
The field of redox-switchable catalysis has gained considerable interest in recent years, enabling catalytic activity and/or selectivity to be oscillated based upon the oxidation-state of the ligand and/or active metal center. Unfortunately, though myriad redox-switchable catalysts have been developed for the ring-opening polymerization of cyclic esters, further fundamental structure–catalytic performance studies are needed to better ascertain how systematic changes in ligand structure impact catalytic activity and redox-switchability. Herein, we describe our studies designed to determine how the number of ligand-based redox-active moieties impacts catalytic performance. More specifically, we compare symmetric catalysts bearing tetradentate [ONNO] ligands with two redox-active moieties to related asymmetric catalysts bearing tridentate [ONN] ligands featuring only a single redox-active moiety. The results of these studies reveal that the number of redox-active moieties may not play a crucial role in the catalysts' switchability; however, the choice of metal center may dramatically influence catalyst activity, stability, and redox-switchability.


 Please wait while we load your content...
Please wait while we load your content...