Rational screening of single-atom-doped ZnO catalysts for propane dehydrogenation from microkinetic analysis†
Abstract
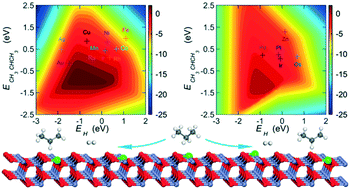
Supported single-atom catalysts, which bridge the gap between homogeneous and heterogeneous catalysis, have attained increasing interest because of their unique catalytic properties and behaviors. In this contribution, periodic DFT+U calculations have been carried out to explore the structural stability, catalytic activity, and selectivity of 13 M1–ZnO (M = Mn–Cu, Ru–Ag, and Os–Au) catalysts in propane dehydrogenation (PDH). Calculated results indicate that the doped single atoms show high resistance to sintering on the ZnO surface. Except on Rh1-, Os1-, Ir1-, and Pt1–ZnO(10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0), a Lewis acid–base interaction occurs, which would greatly enhance the co-adsorption of a pair of amphoteric species on the oxides. The linear chemisorption energy and transition state energy scaling relations hold well for the C–H and H–H bond activation steps involved in the reaction network. Microkinetic analysis is used to determine the activity trend, and volcano-shaped plots of the turnover frequency for propylene production are obtained, with the formation energies of adsorbed H and 2-propyl used as two reactivity descriptors. By using the activation energy difference between propylene dehydrogenation and desorption as a measure of the catalyst selectivity, it is found that a high selectivity can only be achieved at the expense of catalytic activity. Comparison between the energy barriers for H2 desorption and H2O formation indicates that the reduction of ZnO can be suppressed by single-atom doping. The Mn- and Cu-doped ZnO catalysts are suggested to be good catalyst candidates for PDH with lower cost, increased activity, and improved selectivity and catalytic stability.
0), a Lewis acid–base interaction occurs, which would greatly enhance the co-adsorption of a pair of amphoteric species on the oxides. The linear chemisorption energy and transition state energy scaling relations hold well for the C–H and H–H bond activation steps involved in the reaction network. Microkinetic analysis is used to determine the activity trend, and volcano-shaped plots of the turnover frequency for propylene production are obtained, with the formation energies of adsorbed H and 2-propyl used as two reactivity descriptors. By using the activation energy difference between propylene dehydrogenation and desorption as a measure of the catalyst selectivity, it is found that a high selectivity can only be achieved at the expense of catalytic activity. Comparison between the energy barriers for H2 desorption and H2O formation indicates that the reduction of ZnO can be suppressed by single-atom doping. The Mn- and Cu-doped ZnO catalysts are suggested to be good catalyst candidates for PDH with lower cost, increased activity, and improved selectivity and catalytic stability.


 Please wait while we load your content...
Please wait while we load your content...