Interfacial effect of Co4S3–Co9S8 nanoparticles hosted on rGO sheets derived from molecular precursor pyrolysis on enhancing electrochemical behaviour†
Abstract
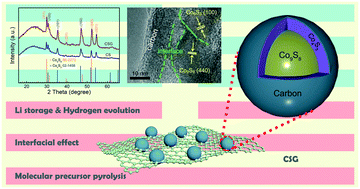
The reasonable design and synthesis of Co-based compounds with superior electrochemical activity and durability have aroused tremendous research interests. Herein, a simple molecular precursor pyrolysis strategy is proposed to fabricate Co4S3, Co9S8 nanoparticles hosted on reduced graphene oxide (rGO) sheets (Co4S3–Co9S8/C/rGO, CSG) using a GO-supported Co-coordination polymer as a precursor for the first time. The synthetic method has the advantages of simple operation and high repeatability. Well-dispersed Co4S3–Co9S8 nanoparticles and abundant interfaces maximize the amount of catalytically active sites. The synergistic adsorption of the reaction intermediates on the interface between Co4S3 and Co9S8 is propitious to electrochemical behavior. The rGO supports with high conductivity and enriched defects speed up the reaction kinetics and effectively inhibit the aggregation of cobalt sulfides. In an alkaline environment, the optimized CSG showed the lowest overpotential (134 mV) at 10 mA cm−2 among the studied materials for the hydrogen evolution reaction. In terms of Li-ion batteries, the hybrid composite CSG exhibited a remarkable reversible capacity of 814 mA h g−1 at a current density of 0.2 A g−1 after 200 cycles. Thus, the present design and engineering strategy may offer an idea for the preparation of other metal sulfides to boost electrochemical storage property.


 Please wait while we load your content...
Please wait while we load your content...