Catalysis to discriminate single atoms from subnanometric ruthenium particles in ultra-high loading catalysts†
Abstract
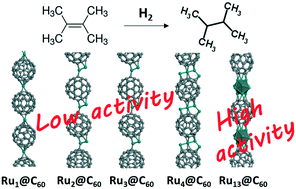
We report a procedure for preparing ultra-high metal loading (10–20% w/w Ru) Ru@C60 nanostructured catalysts comprising exclusively Ru single atoms. We show that by changing the Ru/C60 ratio and the nature of the solvent used during the synthesis, it is possible to increase the Ru loading up to 50% w/w, and to produce hetero-structures containing subnanometric Ru nanoparticles. Several techniques such as high-resolution transmission electron microscopy (HRTEM), scanning transmission electron microscopy-high angle annular dark field (STEM-HAADF), Raman spectroscopy, wide-angle X-ray scattering (WAXS), extended X-ray absorption fine structure (EXAFS) and X-ray photoelectron spectroscopy (XPS) together with theoretical calculations were used to characterize these materials. At such high metal loadings, the distinction between Ru single atoms and clusters is not trivial, even with this combination of techniques. We evaluated the catalytic properties of these materials for the hydrogenation of nitrobenzene and 2,3-dimethyl-2-butene. The catalysts containing only Ru single atoms are much less active for these reactions than the ones containing clusters. For nitrobenzene hydrogenation, this is because electron-deficient Ru single atoms and few atom Run clusters are not performant for H2 activation compared to larger clusters (n ≥ 13), as shown by density functional theory (DFT) calculations. For the more crowded substrate 2,3-dimethyl-2-butene, DFT calculations have shown that this is due to steric hindrance. These simple tests can thus be used to distinguish samples containing metallic subnanometric nanoparticles. These novel catalysts are also extremely active for the hydrogenation of tetra-substituted 2,3-dimethyl-2-butene.


 Please wait while we load your content...
Please wait while we load your content...