Highly selective 1-pentene epoxidation over Ti-MWW with modified microenvironment of Ti active sites†
Abstract
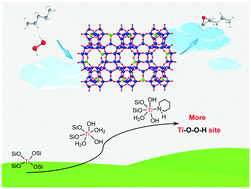
A titanosilicate/H2O2 catalytic system was applied to process the liquid-phase selective epoxidation of 1-pentene to 1,2-epoxypentane (EP). The effects of titanosilicate topology (MWW, MFI, MSE, MEL, MOR, and *BEA), solvent, H2O/H2O2 ratio, catalyst amount, reaction temperature, pressure, and time on the EP production were investigated systematically. The Ti-MWW/H2O2/acetonitrile system exhibited the highest 1-pentene conversion of 72.9% together with high EP selectivity of 99.9% and H2O2 utilization efficiency of 91.5%. Moreover, it was proved that the Ti active sites located inside the intralayer 10-membered ring sinusoidal channels catalyzed the epoxidation process primarily owing to their supplying more steric fitness for 1-pentene molecules. A piperidine (PI)-assisted structural rearrangement of Ti-MWW was performed to further enhance the catalytic activity, almost doubling the turnover number value. The evolution of the microenvironment of Ti active sites in this structural rearrangement process was carefully investigated, revealing the coordination of N atoms in PI molecules to the Ti atoms. More importantly, we identified that the hexa-coordinated Ti sites with the PI molecules as ligand could significantly accelerate H2O2 activation, the effect of which far exceeded the inhibition effect caused by the electronegativity increase of Ti active sites.


 Please wait while we load your content...
Please wait while we load your content...