Strong metal–support interactions in Pd/Co3O4 catalyst in wet methane combustion: in situ X-ray absorption study†
Abstract
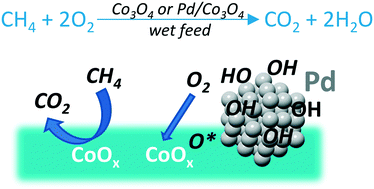
Co3O4 and Pd/Co3O4 catalysts were analyzed for Pd and Co speciation during wet lean methane combustion in temperature ranges of 150–450 °C by means of in situ X-ray absorption spectroscopy. As water inhibits Pd reoxidation during methane combustion, the catalysts were reduced prior to the in situ XAS analyses to follow the evolution of the oxidized species. The contributions from metallic Pd, PdO and Pd(OH)2 and metallic Co, Co(II), and Co(III) oxides were quantified as a function of temperature. The Pd-free sample showed remarkable stability of its species regardless of temperature in the dry feed but the presence of water inhibited its reoxidation. In a partially pre-reduced Pd/CoOx catalyst in the wet feed, cobalt species progressively oxidized to the level of the Pd-free sample. Pd remained largely unoxidized below the surface, with the Pd(0) fraction remaining stable in the studied 150–450 °C region. This was ascribed to the effect of the cobalt oxide support, as Pd(0) is known to oxidize progressively for alumina and tin dioxide supports in the wet feeds. We considered bulk thermodynamics of the involved processes, as well as the reported activation energies and kinetic data from a previous study on methane combustion. In combination with the results from the in situ XAS, we suggest that cobalt oxide, being a preferential oxygen but unlikely hydroxyl acceptor, inhibits Pd oxidation, and does not provide benefits in terms of inactive Pd(OH)2 formation.


 Please wait while we load your content...
Please wait while we load your content...