Sn4+ and S2− co-doped N–TiO2−x nanoparticles for efficient and photocatalytic removal of contaminants†
Abstract
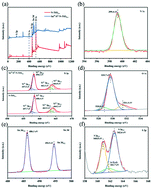
N–TiO2−x nanoparticles (NPs) were selected as a basic construction unit because there are many Ti3+ and O vacancies on its surface. When S2− and Sn4+ are co-doped into the N–TiO2−x lattice, the O2− and Ti4+ atoms can be replaced, respectively. The formation of dopant energy levels from Sn4+ and S2− in Sn4+/S2−/N–TiO2−x causes an obvious red shift to the visible light region. The recombination rate of the e−/h+ pairs was significantly reduced due to the fast migration and separation of the electrons as a result of the energy level doping from Sn4+ and S2− and the formation of electron traps with Sn4+. The N–TiO2−x NP band gap was significantly narrowed from 3.07 eV to 0.77 eV upon co-doping with Sn4+ and S2−, resulting in the superior photocatalytic activity of the Sn4+/S2−/N–TiO2−x NPs. The photocatalytic reduction rate of Cr(VI) increased by 11.6 times and the photodegradation rate of doxycycline hydrochloride (DH) achieved about 5.7 times that of the original N–TiO2−x NPs. This study provided a simple approach to preparing metal and non-metal co-doped N–TiO2−x NPs, which could greatly improve the removal efficiency of contaminants via their synergistic effect.


 Please wait while we load your content...
Please wait while we load your content...