Enhancing aqueous pollutant photodegradation via a Fermi level matched Z-scheme BiOI/Pt/g-C3N4 photocatalyst: unobstructed photogenerated charge behavior and degradation pathway exploration†
Abstract
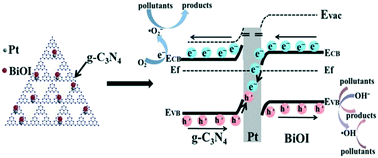
A Z-scheme photocatalytic system has been widely called for to solve water pollution problems. However, the mechanism for the degradation of pollutants with a Z-scheme photocatalyst has so far been ambiguous. In this work, an efficient all solid-state BiOI/Pt/g-C3N4 photocatalyst was conceived and fabricated based on the Fermi level matched principle. The morphology, structure and chemical composition were characterized in detail. The degradation rate for phenol over as-prepared BiOI/1% Pt/g-C3N4 was 6.0, 2.3 and 1.8 times higher than those of pristine g-C3N4, BiOI/g-C3N4 and Pt/BiOI, respectively. Furthermore, the highest degradation rate constant (0.0514 min−1) for tetracycline hydrochloride (TCH) could be achieved with BiOI/1% Pt/g-C3N4. Notably, the outstanding degradation performance was ascribed to unobstructed photogenerated charge separation and transfer resulting from the appropriate interfacial interaction of Pt/g-C3N4 and BiOI/g-C3N4, which was verified by the results of electron spin resonance studies, reactive oxidation species scavenger experiments, and work function analysis. Characterization results from fresh and used photocatalyst samples, and cyclic degradation and toxicity experiments demonstrated the extremely high stability and low toxicity of BiOI/1% Pt/g-C3N4. Accordingly, the comprehensive degradation mechanism based on photophysical and photochemical processes was investigated in depth. The rationally designed all solid-state photocatalyst has prospects for environmental remediation.


 Please wait while we load your content...
Please wait while we load your content...