An in situ grown lanthanum sulfide/molybdenum sulfide hybrid catalyst for electrochemical hydrogen evolution†
Abstract
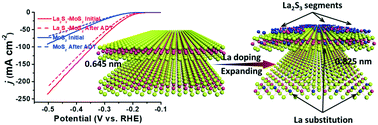
Hydrogen has great promise as an energy carrier superior to traditional fossil fuels. Electrochemical water splitting is expected to be an effective and sustainable method for producing high-purity hydrogen fuel cleanly. The hydrogen evolution reaction (HER) is the cathodic reaction in the electrochemical water-splitting process and requires efficient catalysts. However, the state-of-the-art Pt suffers from high cost and rare reserves, which significantly limit its extensive applications in the hydrogen evolution reaction for water splitting. Although its catalytic activity still falls far short of expectations, molybdenum sulfide has emerged as one of the most promising catalysts for HER due to its abundant reserves and high tunability. Here, we have reported the synthesis and HER catalytic performance of a hybrid lanthanum sulfide/molybdenum sulfide (La2S3–MoS2) catalyst. The formation of molybdenum sulfide nanosheets in situ with lanthanum sulfide deposition was achieved through a facile one-pot solvothermal strategy. The present catalyst displayed much superior activity for HER to pristine and commercial molybdenum sulfide; low overpotentials of 205 mV, 277 mV, 336 mV and 455 mV for the current densities of 10 mA cm−2, 50 mA cm−2, 100 mA cm−2 and 200 mA cm−2 were obtained, respectively, together with a low Tafel slope of 51 mV dec−1. Moreover, the present catalyst exhibited excellent stability in long-term hydrogen evolution. The remarkable HER performance might be mainly due to the expanded interlayer distance and the nano-interfaces between the molybdenum sulfide and lanthanum sulfide segments, which induced an increased exposure of active sites and a modified surface electronic structure.


 Please wait while we load your content...
Please wait while we load your content...