From cellulose to 1,2,4-benzenetriol via catalytic degradation over a wood-based activated carbon catalyst†
Abstract
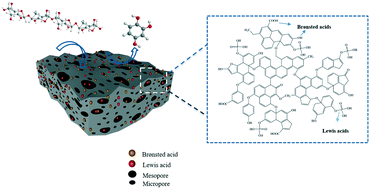
Spherical P-doped activated carbon (CSP) was obtained from poplar bark by microwave carbonization with phosphoric acid as the dopant and activating agent. Phosphorus bonded with four oxygen atoms to form a phosphate-like structure rather than directly bonding with C atoms in CSP; such groups can provide Lewis acid active sites with moderately strong acidity. The Lewis acid and total acid content increased with increasing phosphoric acid doping ratio. The use of phosphoric acid as an activating agent promotes the formation of a developed porous structure. CSP1 (mass ratio of raw material to acid = 1 : 1) had a specific surface area of 1115 m2 g−1 with developed micro–mesoporous structures, mainly micropores. Investigation of the activity of CSP in catalytic cellulose hydrolysis during hydrothermal degradation showed that Brønsted acid sites promote hydrolysis and dehydration of cellulose, Lewis acid sites promote subsequent reactions such as isomerization, rearrangement, and oligomerization, and the developed pores can provide reaction sites and facilitate reactant transportation. The optimal reaction conditions, i.e., 1 h at 230 °C and 1 MPa with CSP1 (0.4 g) as the catalyst, gave a cellulose conversion of >99.9%, with 42.30% selectivity for 1,2,4-benzenetriol. This study provides an effective method for preparing 1,2,4-benzenetriol from cellulose with biomass-based activated carbon as the catalyst, which enables green synthesis of 1,2,4-benzenetriol and promotes the use of biomass waste resources.


 Please wait while we load your content...
Please wait while we load your content...