In situ synthesis of ultrafine metallic MoO2/carbon nitride nanosheets for efficient photocatalytic hydrogen generation: a prominent cocatalytic effect†
Abstract
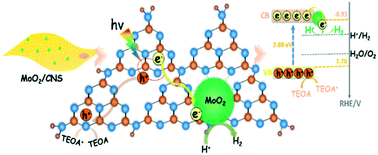
The development of noble metal free and highly active cocatalysts is critically important and also challenging for the practical applications of photocatalytic hydrogen production. Here, ultrafine MoO2 cocatalyst nanoparticles (≤10 nm) were in situ solvothermally synthesized on carbon nitride nanosheets (CNSs). We found that the ultrafine MoO2 nanoparticle decorated CNS exhibits higher electrocatalytic hydrogen evolution reaction (HER) performance than Pt–CNS. Besides, CNS with a curly thin nanosheet structure has an increased specific surface area and broadened bandgap after the solvothermal process compared to bulk carbon nitride (CN). Characterization results reveal that the strong interfacial interaction between metallic MoO2 and CNS greatly promotes the photogenerated electrons extracted from CNS to MoO2, resulting in efficient electron excitation and charge transfer in the composite. Accordingly, the hydrogen production rate of 1.0 wt% MoO2/CNS is 3.4 and 1.6 times higher than that of the same mass noble-metal Pt loaded CN and CNS under visible light irradiation, respectively. This work may provide a guide for constructing non-noble metal cocatalyst modified materials for efficient photocatalytic hydrogen production.


 Please wait while we load your content...
Please wait while we load your content...