Development of BiOI as an effective photocatalyst for oxygen evolution reaction under simulated solar irradiation†
Abstract
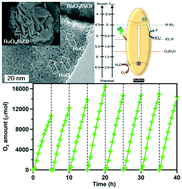
In this study, crystalline BiOI powders were prepared for photocatalytic O2 evolution in the presence of NaIO3 as the electron mediator. BiOI with a microspherical morphology, a layered structure composed of [Bi2O2]2+ and intercalated I− ions, exhibited a suitable valence band level to generate photoexcited holes for O2 evolution. Moreover, ruthenium was loaded using the impregnation or photodeposition method to produce RuO2 as a co-catalyst to improve the photocatalytic activity of BiOI. Photodeposited RuO2-loaded BiOI showed a high O2 evolution rate of 2730 μmol h−1 and can be reused eight times in the presence of NaIO3 under simulated solar irradiation. The high photocatalytic O2 evolution can be attributed to the highly dispersed RuO2, which could serve as an effective electron sink, on the surface of BiOI and its enhanced visible light-harvesting ability. Besides, the presence of NaIO3 in the system was effective to receive photoexcited electrons from RuO2-loaded BiOI for improving charge separation and hence the O2 evolution from RuO2 sites on the BiOI surface. The RuO2-loaded BiOI with high photocatalytic activity and stability for generating O2 could be a potential candidate for achieving overall water splitting in a Z-scheme system in the presence of NaIO3 for solar utilization in the future.


 Please wait while we load your content...
Please wait while we load your content...