The role of boron sites in side-chain alkylation of toluene with methanol and a high performance composite catalyst†
Abstract
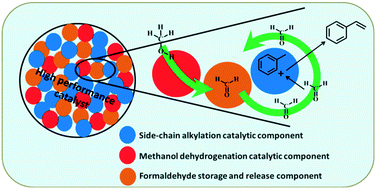
Side-chain alkylation of toluene with methanol has attracted interest for decades as a one-step alternative pathway to manufacture styrene under mild conditions, and a high performance catalyst is most urgently needed to realize the commercial application of this technology. Herein, we studied the role of boron sites in improving the styrene/ethylbenzene ratio (ST/EB) in the products primarily. It was found that the introduction of boron sites into cesium ion exchanged zeolite X (CsX) is helpful in adsorbing and stabilizing the generated formaldehyde from methanol dehydrogenation over the base sites of CsX, and this mainly accounts for the inhibition of further conversion of styrene into ethylbenzene in the side-chain alkylation products. Moreover, it was found that the catalytic performance of the CsX-based catalyst with boron sites was influenced tremendously by the distance between the boron sites and the base sites of CsX. Introducing boron sites into the CsX-based catalyst by grinding boric acid impregnated SiO2 (B/SiO2) and CsX was an efficient way to greatly improve the ST/EB in the side-chain alkylation products. Finally, based on the above findings, a ternary composite catalyst with a high yield of side-chain alkylation products (styrene and ethylbenzene) and high ST/EB, which were 39.45% and 1.81, respectively, was developed by mixing CsX, cesium oxide modified CsX and B/SiO2 with the grinding route. In addition, the key points for preparing a more efficient catalyst in the future were also discussed and proposed.


 Please wait while we load your content...
Please wait while we load your content...