Hourglass-type polyoxometalate-based crystalline materials as efficient cooperating photocatalysts for the reduction of Cr(vi) and oxidation of dyes†
Abstract
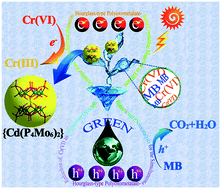
The visible-light-driven simultaneous conversion of toxic Cr(VI) and organic dyes is a promising approach to alleviate environmental pollution, in which the key point is developing efficient photocatalysts. Here, a series of reduced MoV-contained hourglass-type polyoxometalate crystalline materials with a formula (H2bpp)2[{Na4(H2O)5}{Co0.8Cd0.2(H2O)2}{Cd[Mo6O12(OH)3(H2PO4)(HPO4)(PO4)2]2}]·2H2O (1), (H2bpp)3[Zn2{Cd[Mo6O12(OH)3–(HPO4)3(PO4)]2}]·6H2O (2), and [(H2bpp)3(bpp)2][{Al2(H2O)4}{Cd[Mo6O12(OH)3(H2PO4)2(HPO4)(PO4)]2}2]·9H2O (3) (bpp = 1,3-bi(4-pyridyl)propane) were prepared as synergetic photocatalysts for Cr(VI) reduction and methylene blue (MB) oxidation under visible-light irradiation. The three assemblies display wide visible-light absorption ranges, suitable energy-level structures and excellent redox activity. When used as photocatalysts, assemblies 1–3 exhibited excellent visible-light photocatalytic activity. Under the original pH conditions, the removal ratios of Cr(VI) can reach 74%, 68%, and 55%, and the degradation ratios of MB are 96%, 87%, and 88% for 1–3 with 3 hours irradiation, respectively. These results were higher than those of the separate half reactions, suggesting that the synergistic effect between Cr(VI) reduction and MB oxidative degradation accelerates the separation and utilization of photogenerated electrons and holes. The possible reaction mechanism has also been provided. This work provides some guidance for reduced polyoxometalate-based materials as potential candidates for environmental restoration.


 Please wait while we load your content...
Please wait while we load your content...