The direct decomposition of NO into N2 and O2 over copper doped Ba3Y4O9†
Abstract
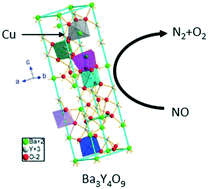
The direct decomposition of nitrogen oxides (NOX) is an ideal way to remove NOX pollution created by chemical industries and automobiles. Ba3Y4O9 is a promising candidate for the NOX direct decomposition reaction, however, its catalytic activity is not high enough and the stability is insufficient. Here, NO decomposition activity on several metal ion doped Ba3Y4O9 catalysts is reported and it is found that Cu is the most effective for increasing the activity. With 10% Cu doping, the catalytic activity and the stability were improved simultaneously. Temperature programmed desorption (TPD) results indicate that doping with Cu increased the amount of surface oxygen vacancies and mobility of the lattice oxygen resulting in an increase of NO adsorption and the promotion of the decomposition of NOX intermediate species. In situ FT-IR spectra prove that Cu doping weakened the association of NO3− species, thus promoting the decomposition of NO3− species at low temperatures, which resulted in high catalytic activity for NOX decomposition. These results indicate that introducing Cu into the Ba3Y4O9 system is an effective way to achieve high activity and a stable catalyst for the direct decomposition of NOX.


 Please wait while we load your content...
Please wait while we load your content...