Identifying and controlling the acid site distributions in mordenite zeolite for dimethyl ether carbonylation reaction by means of selective ion-exchange†
Abstract
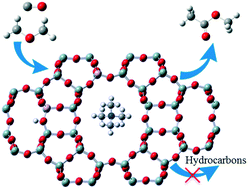
As Brønsted acid sites in different types of channels exhibit distinct catalytic behaviors in the dimethyl ether (DME) carbonylation reaction over acidic mordenite (H-MOR) zeolites (e.g. acid sites in 8-membered ring channels for carbonylation reaction, acid sites in 12-membered ring channels for methanol-to-hydrocarbons reactions), the identification and regulation of acid site distribution in mordenite zeolites are of great importance to improve the catalytic performance. In this work, we employ the selective ion-exchange method to identify and control the acid site distribution in mordenite zeolites, and the chemical properties of acid sites in different channels are investigated by NH3-TPD and CD3CN FT-IR. Additionally, the effect of selective ion-exchange on the catalytic performance of DME carbonylation reaction is also discussed. The selective ion-exchange is realized by using tetramethylammonium (TMA+) ions, which can selectively remove the counter ions in the 12-membered ring channels but are inaccessible to the counter ions in 8-membered ring channels due to the steric hindrance. The selective ion-exchange reveals that the relative amounts of acid sites in 12-membered ring channels and 8-membered ring channels are 68% and 32%, respectively. Interestingly, it is found that introducing TMA+ into H-MOR zeolites significantly improves the catalytic activity and stability in DME carbonylation reaction as a result of depressing the methanol-to-hydrocarbons reactions. The 3TMA-H-MOR catalyst shows high stability for 210 hours on stream. The present work opens a new avenue for designing carbonylation catalysts with excellent stability in DME carbonylation reaction.


 Please wait while we load your content...
Please wait while we load your content...