Carbon deposits during catalytic combustion of toluene on Pd–Pt-based catalysts†
Abstract
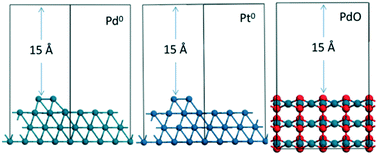
Carbon is easily deposited on a catalyst during catalytic combustion of toluene, resulting in catalyst deactivation. In this paper, a series of Pd–Pt-based mesoporous catalysts were prepared using an incipient wetness impregnation method, and the effects of the catalyst pore size, acidity, and noble metal loading, the chemical state of palladium and the oxygen supply performance of palladium species on carbon deposition during the catalytic combustion of toluene were investigated. The in situ transmission infrared test was used to study the intermediate products of the catalytic combustion of toluene on Pd–Pt-based catalysts. The results show that CO was produced during the catalytic combustion of toluene on Pd–Pt-based catalysts and adsorbed onto the Pd0 phase by bridge adsorption. Carbon formed via the disproportionation reaction of CO (2CO → C + CO2) on the Pd0 phase was deposited onto the catalysts, leading to carbon deposition. The addition of Pt and CeO2 increased the content of the PdO phase on the catalysts and the oxygen supply performance of the palladium species, thereby promoting the oxidation of CO to CO2, reducing the disproportionation reaction of CO on the Pd0 phase, and inhibiting the formation of carbon deposits.


 Please wait while we load your content...
Please wait while we load your content...