A dinuclear iron complex as an efficient electrocatalyst for homogeneous water oxidation reaction†
Abstract
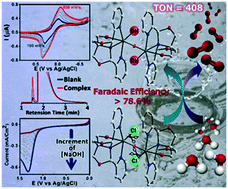
Among the current technologies available for producing the environment friendly fuel hydrogen, the water splitting reaction has attracted immense attention. However, such a vital reaction has its own drawbacks in the form of compromised efficiency and the turn-over number of water electrolysis. To counteract these drawbacks, the development of efficient electrocatalysts for the OER is highly expedient. In this present work the electrocatalytic activity of a newly synthesized dinuclear oxo-bridged iron complex [(FeLCl)2O](FeCl4)2 (L = (2-(pyrridin-2-yl)oxazolidine-4,4-diyl) 1 is unveiled. The electrocatalytic activity towards water oxidation reaction has been determined in organic medium with added NaOH. Experimental results show that catalysts are highly stable and molecular oxygen is produced via electrochemical water oxidation with an appreciable turnover number (408) coupled with a high faradaic efficiency (>78.6%). Mechanistic investigation revealed that the replacement of chlorides from complex 1 by hydroxide ions helps in the O–O bond formation during the process of water oxidation, which represents the most crucial step for the oxidation of water to molecular oxygen and this was further substantiated by ESI-MS measurements and UV-VIS studies. To explain such a typical mechanism, DFT calculations were performed. Furthermore, to prove the exclusive role of the cationic part of complex 1, we have synthesized another control complex with a different counter anion [(FeLCl)2O]Cl22 and used it in the electrocatalytic water oxidation reaction. From the results, it was found that control complex 2 has the same efficacy as complex 1.


 Please wait while we load your content...
Please wait while we load your content...