Effects of Ni loading on the physicochemical properties of NiOx/CeO2 catalysts and catalytic activity for NO reduction by CO
Abstract
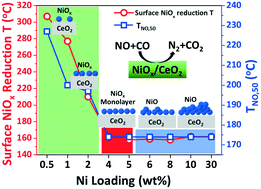
Transition metal oxide catalysts have been investigated extensively because of their relatively low cost and high activity in many chemical reactions. In this work, a series of NiOx/CeO2 catalysts (0.5–30 wt% Ni) were prepared using the incipient wetness impregnation method. These catalysts were tested with various characterization techniques, including Brunauer–Emmett–Teller (BET) theory, Raman spectroscopy, X-ray powder diffraction (XRD), hydrogen temperature-programmed reduction (H2-TPR) and gas chromatography (GC) for their physicochemical properties, surface properties, reduction properties and catalytic activities in the NO reduction by CO reaction. The increase in Ni loading of the catalyst (up to 5% NiOx/CeO2) led to the decrease in specific surface area, formation of NiOx crystalline structures on the CeO2 surface, easier reduction of the catalyst compared to bulk NiOx and bulk CeO2, and increase in catalytic activity in the NO reduction by CO reaction. From these results, the surface dispersion of NiOx and the formation of monolayer NiOx coverage of the catalysts were believed to affect the catalytic activity greatly. The results provided insights into the structure–activity relationship of NiOx/CeO2 catalysts for the NO reduction by CO reaction.


 Please wait while we load your content...
Please wait while we load your content...