In situ addition of Ni salt onto a skeletal Cu7S4 integrated CdS nanorod photocatalyst for efficient production of H2 under solar light irradiation†
Abstract
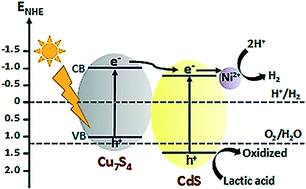
Development of earth-abundant, low cost, skeletal-type copper sulfide superstructures plays a prominent role in various potential applications, owing to the properties of superstructures such as excellent permeability of charge and mass. In this study, we synthesized hollow superstructures of crystallinity-controlled polyhedral skeletal-type copper sulfide (Cu7S4), which were integrated with cadmium sulfide (CdS) nanorods. The photocatalytic hydrogen production by water splitting was investigated under solar light irradiation with lactic acid as a hole scavenger. The Cu7S4/CdS composite exhibited a H2 production rate of 6.25 μmol h−1, which was 3 times higher than that of CdS. Moreover, the addition of a small amount of nickel salts to the reaction solution significantly improved the activity of Cu7S4/CdS for H2 production (45 μmol h−1), which was 20 times higher than that of CdS and 7 times higher than that of Cu7S4/CdS. The enhancement of activity in Ni–Cu7S4/CdS (Cu7S4/CdS with Ni salts in the reaction solution) was attributed to the effective separation of charge carriers due to the unique properties of the skeletal-type Cu7S4 cages and the superior charge transportation of Ni salts in the reaction solution. The Cu7S4/CdS system containing Ni salts in the reaction solution is reported for photocatalytic hydrogen production for the first time, to the best of our knowledge, which promises new and inspiring research for the development of photocatalytic water splitting applications.


 Please wait while we load your content...
Please wait while we load your content...