Probing into the effect of heterojunctions between Cu/Mo2C/Mo2N on HER performance†
Abstract
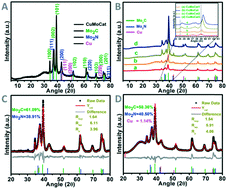
Hydrogen is one of the cleanest forms of energy and can solve several issues, including environmental pollution and depletion of fossil fuels. The hydrogen evolution reaction (HER), being a carbon-neutral process, can reduce the amount of carbon in the earth's atmosphere. Molybdenum based solids are among the most popular electrocatalysts, and have been explored extensively for HER processes as an alternative to platinum. Herein, we report a nanostructured electrocatalyst (CuMoCat) consisting of molybdenum carbide, molybdenum nitride, and elemental copper, forming a heterojunction within the composite. Copper doping into a molybdenum carbide/nitride composite not only facilitates the lowering of the overpotential but also enables the catalyst to perform at high current density. CuMoCat exhibits an overpotential of 82 mV for attaining a current density of 10 mA cm−2 with a Tafel slope value of 33 mV dec−1 and shows excellent stability of 3000 cycles in acidic media. A theoretical study reveals that Cu doping brings a change in the electronic properties of the catalyst, which improves the overall adsorption and desorption of hydrogen on the catalyst surface during the hydrogen evolution reaction. The free energy diagram for the ‘Mo’ and ‘N’ hydrogen adsorption sites of CuMoCat shows a shift of free energy making these more favourable for the HER with respect to the Mo/N sites of the un-doped catalyst.


 Please wait while we load your content...
Please wait while we load your content...