Sonochemical engineering of highly efficient and robust Au nanoparticle-wrapped on Fe/ZrO2 nanorods and their controllable product selectivity in dimethyl oxalate hydrogenation†
Abstract
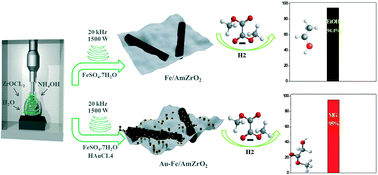
Considerable attention has been paid in last few years to the environmentally-benign hydrogenation reaction of dimethyl oxalate (DMO) to produce methyl glycolate (MG), ethylene glycol (EG) and ethanol (EtOH). Cu-Based composites and noble-metal promoted Cu-materials have been reported as highly efficient catalysts in DMO hydrogenation. In this study, for the first time, a Cu-free Fe/ZrO2 nanorod catalyst with a highly crystalline structure was synthesized in the absence of any toxic materials and through a one-pot sonochemical approach and used without the calcination step as a robust catalyst in DMO hydrogenation to produce a high EtOH yield of 94.4%. Furthermore, to control the product distribution in this multistep DMO hydrogenation and achieve a high MG selectivity of 95%, highly dispersed Au nanoparticles were nicely decorated on the surface of the Fe/ZrO2 nanorod catalyst by a facile sonochemical route which was used as a highly efficient catalyst. Moreover, the produced catalysts displayed high stability properties for a period of at least 300 h with maintained high catalytic activity. Interestingly, X-ray diffraction results for the spent catalysts demonstrated that Hägg iron carbide (Fe5C2) is the active phase for the production of ethanol using the Fe/ZrO2 catalyst, while the XPS results exhibited that the co-existence of the three oxidation states of metallic Au (Au+, Au0, and Au3+) plays the key role as the active species for the synthesis of MG with high selectivity using the Au/Fe/ZrO2 catalyst.


 Please wait while we load your content...
Please wait while we load your content...