PtRu/Zn3Ce1Ox catalysts with Lewis acid–base pairs show synergistic performances for the conversion of glycerol in the absence of externally added H2†
Abstract
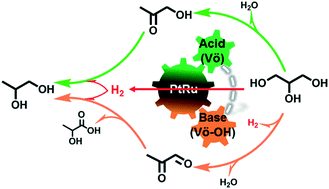
Previous studies revealed that tandem dehydration, dehydrogenation and hydrogenation, promoted by metal–support interfacial catalysis, play a critical role in determining the intrinsic kinetics of transfer hydrogenolysis of bio-oxygenates. However, the synergism of metal and acid–base pairs for tunable C–H, O–H and C–O cleavage and metal–H strength is under debate in this area. Herein, we present a series of bimetallic PtRu/Zn3Ce1-LDO catalysts, with abundant Lewis acid–base pairs and heterojunction structures at the metal–support interface, exhibiting enhanced performances of transfer hydrogenolysis of glycerol. The key finding is that the incorporation of ZnO unexpectedly induced the formation of large amounts of oxygen vacant sites and surface hydroxyl sites on the CeOx support, leading to abundant Lewis acid–base pairs. The strong electron coupling effect of PtRu and Lewis acid–base pairs results in weakened PtRu–H bonding, facilitating a hydrogen transfer reaction. Synergism between enhanced Lewis acid–base pairs and weaker PtRu–H hydride bonding is critical for remarkable catalytic activity (TOF: 526 h−1) and selectivity towards glycols (70.8%), and results in one of the best performances in the current literature. Moreover, the influence of hydrogen donors, reaction temperature and time on conversion and product distribution was further investigated in detail. An alternative reaction pathway for the transfer hydrogenolysis of glycerol over the proposed acid–base pair catalysts was proposed and validated with experimental data. The outcome of this work will provide new insights into the rational design of efficient catalytic materials for energy and environmental applications.


 Please wait while we load your content...
Please wait while we load your content...