Exploring CoP core–shell nanosheets by Fe and Zn dual cation doping as efficient electrocatalysts for overall water splitting†
Abstract
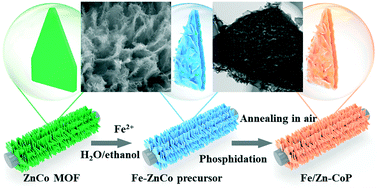
Dual cation doping engineering can achieve high-performance electrocatalysts through the synergic effects between the two cation dopants. Here, we report a dual cation (Fe, Zn)-doped CoP core–shell nanostructure (Fe/Zn-CoP) derived from a metal–organic framework as a robust electrocatalyst. In this strategy, the introduction of Fe not only changes the morphology to provide abundant active sites, but also modifies the electronic structure of CoP to improve intrinsic activity. Furthermore, the Zn element as a sacrificial dopant can expose more active sites to boost the catalytic reaction. Consequently, the Fe/Zn-CoP core–shell arrays exhibit low overpotentials of 75 mV for the hydrogen evolution reaction (HER) and 267 mV for the oxygen evolution reaction (OER) at 10 mV cm−2 in an alkaline medium. When employed as both the anode and the cathode, Fe/Zn-CoP needs a low cell voltage of only 1.59 V to achieve 10 mV cm−2. This work highlights a feasible method for combining nanostructure design and dual cation doping to prepare highly efficient electrocatalysts.


 Please wait while we load your content...
Please wait while we load your content...